BioSignature review: Are hormones the key to weight loss?
There’s a story behind this article. It was originally published in Dutch on the blog of Guy Droog, a Men’s Health cover model and personal trainer who was a long term client of mine. Guy and I share a commitment to the truth even when this upsets many people and probably isn’t a wise move careerwise. This particular article instigated such a controversy that the owners of Overload Worldwide got involved. Overload Worldwide is an organization that preaches the sayings of Charles Poliquin and makes major bank in the process of certifying others to do the same. As did tons of other personal trainers that were certified as Personal Hormonal Profiling (PHP) trainers. They challenged Guy to a debate. Guy and I have a clear conscience and are always open to challenge our views, so we both accepted. Radio silence. Rob Trousselot, the owner of LIJFSTIJL Coaches, picked up on the story and invited Overload Worldwide, Guy, me and Chi L. Chiu, representative of Chivo, a scientific health institute, to a public debate at his cost. Guy, me and Chi accepted the invitation. To this day we have not heard back from Overload Worldwide.
A Scientific Review of Charles Poliquin’s BioSignature Modulation
Stubborn fat. Women can’t get rid of the fat on their hips that leaves them with a fat ass and men can’t get rid of the belly that obscures the glorious six-pack underneath. Fear not, there is a solution. The distribution of fat on your body is determined by your hormonal balance. Fix your hormonal balance and your stubborn fat will disappear like tears in a blizzard. The best part is you don’t even have to eat less.
So goes the story of an exponentially rising number of organizations that promise a perfect beach body by correcting your hormonal imbalances, notably Charles Poliquin’s BioSignature Modulation and Overload Worldwide’s Personal Hormonal Profiling. How scientific are these theories and how useful is their application? This article focuses on BioSignature, because it’s the grand daddy of them all.
The essence of BioSignature is as follows. You can determine the serum concentrations of the most important hormones in your body without blood work. You do this by measuring your skinfold thickness on various sites on your body using calipers. Your regional fat storage at specific sites is indicative of specific hormones being out of whack. For example, if you store a disproportionate amount of fat on the subscapular site (below your shoulder blades), this indicates that you produce too much insulin. When you know which hormones are unbalanced, you can correct this using specific training, nutrition, supplementation and lifestyle interventions. The result is that your undesirable fat storage pattern will correct correspondingly.
This image summarizes the BioSignature Modulation hormonal assessment. The next one shows Personal Hormonal Profiling, which is virtually identical.
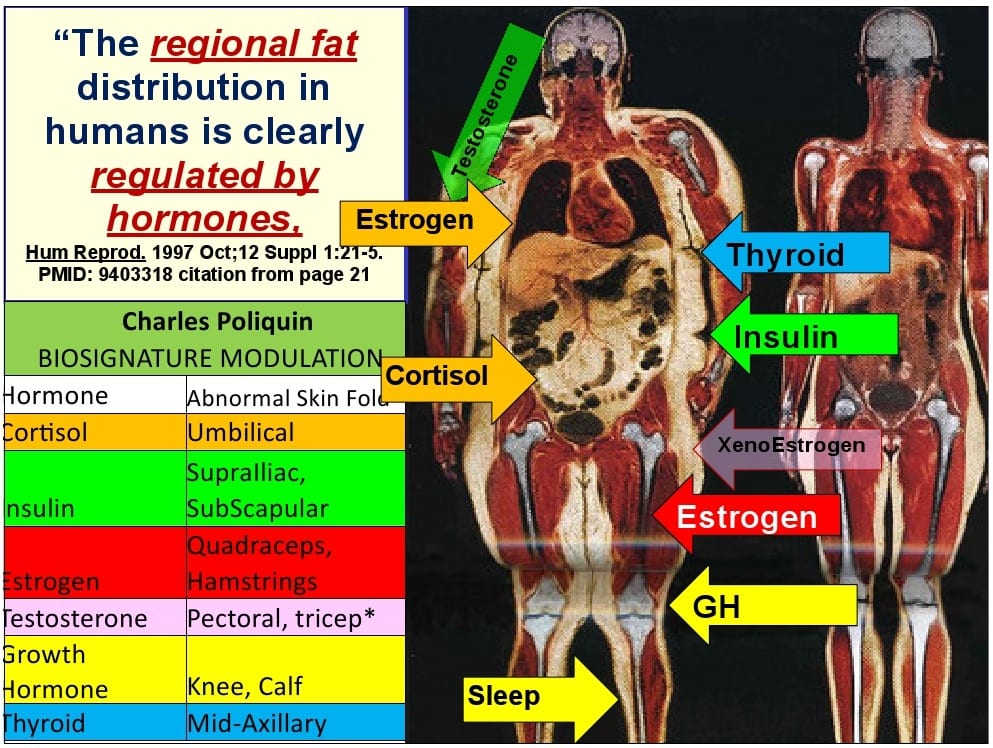
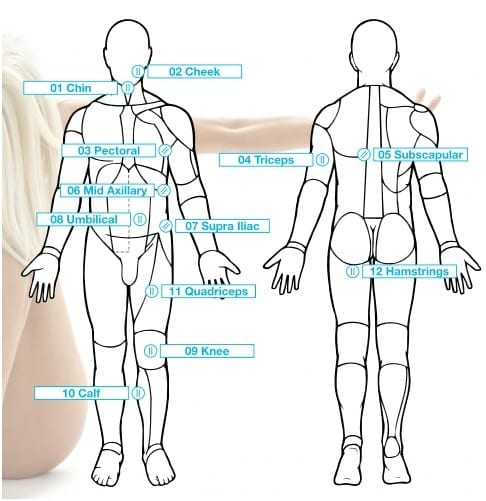 BioSignature/Personal hormonal profiling: based on 12 skinfold thickness measurements your hormonal balance is determined.
BioSignature/Personal hormonal profiling: based on 12 skinfold thickness measurements your hormonal balance is determined.
Divide and conquer stay fat
Before going into any research we can already address a major logical problem of BioSignature. Your fat distribution and your fat mass are two distinct anthropometrical (literally ‘bodily measurement’) characteristics.
- Fat mass is the total amount of fat on your body
- Fat distribution defines where fat mass is stored.
If you change your fat distribution via hormonal correction without actually losing fat, you’re simply shifting the fat around your body. For example, during menopause women ‘lose’ fat on their hips and legs due to hormonal changes. This fat relocates primarily to the abdomen [2]. The result is a flat ass and a gut. Not exactly the magic solution to fat loss.
A grain of truth
Redistribution of your fat mass is thus not always desirable. But sometimes it is. Most men wouldn’t mind having the fat on their abs shifted to their legs. Can this be done by changing your hormonal balance? It most definitely can. BioSignature is not something Poliquin made up out of thin air. The scientific literature is unequival about the fact that your regional fat distribution is affected by the activity of various hormones [2].
Yet this is not the end of the hormone story. How your hormones affect localized fat storage is not as BioSignature claims.
The sex hormones
Most people are familiar with the folk wisdom that men have an apple shape, i.e. relatively much visceral and intra-peritoneal fat storage, and women have a pear shape, i.e. relatively much gluteo-femoral fat storage. The following picture from a scientific journal article demonstrates this.
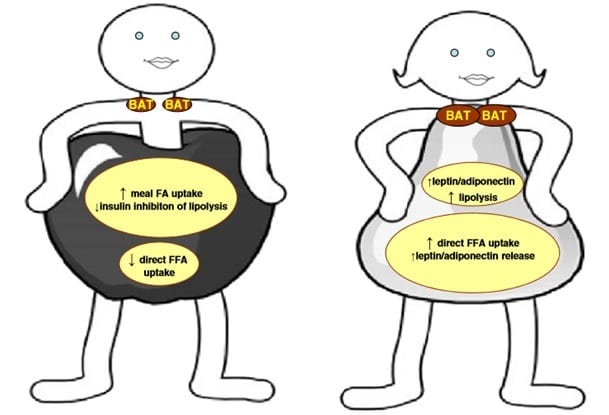
The gender difference in fat storage pattern is a fact, as everyone can observe in daily life. It is easy to think that this is a result of men having high testosterone levels and women having high estrogen levels. Those are after all the hormones involved in sexual differentiation.
However, this is a typical case of mistaking correlation for causality. The truth is that both differences in hormone production and differences in fat storage pattern are largely genetically determined. Approximately 50% of the difference in fat storage pattern between men and women can be explained solely by our DNA. Within a gender it’s even 70% [2]. I’ll revisit transsexuality in relation to fat distribution later on, but for now I’ll assume that people are not interested in a sex change simply to ‘balance’ their hormonal profile.
Of the remaining 30% of the difference in fat distribution between individuals of the same gender, age and ethnicity explain the majority [2]. This already indicates that the role of hormones in influencing fat storage pattern is marginal compared to the gross differences we can observe between individuals. However, as I noted earlier, at the individual level, hormones can still make a great difference in a person’s fat distribution.
Testosterone
Let’s start with reviewing testosterone, because I don’t do ‘ladies first’. I don’t put people on a pedestal simply because they have boobs. In men, even injecting up to 10 times supraphysiological doses of testosterone or shutting down testosterone production entirely does not alter where the body stores fat. Testosterone affects the depth of the fat storage but not on which body part its stored, so you cannot tell what a man’s testosterone level is by looking at his fat storage pattern like BioSignature does.
If testosterone did influence regional fat distribution, you’d expect men and women to store fat in the exact opposite way they actually do. Testosterone acts via the androgen (‘man making’) receptor and stimulates lipolysis AKA fat burning. Androgen receptor density is much higher in the upper than the lower body, so this would imply that men store fat primarily in their lower bodies and women store fat primarily in their upper bodies [2]. Fortunately, this is not the case and men don’t have female curves.
Stress
Testosterone can affect where you store fat indirectly and this is where it gets complex. Testosterone combats the effects of cortisol, the stress hormone.
Cortisol is the hormone with the strongest relation to fat distribution in both genders. Cortisol acts via the glucocorticoid receptor to stimulate lipoprotein lipase (LPL). Simply put, LPL is an enzyme that makes fat cells ready for storage. The notorious fat in between the organs in your abdomen consists mainly of fat cells called visceral adipocytes. Visceral adipocytes have more glucocorticoid receptors than other fat cells. Fat cells in the torso have fewer glucocorticoid receptors and subcutaneous fat cells found on the rest of your body have even fewer. The fat cells on your hips and upper legs have the least glucocorticoid receptors.
This means cortisol causes fat storage primarily on your abdomen, followed by the rest of your torso and only minimally on your lower body. The claim of BioSignature that lots of abdominal fat storage is an indicator of high cortisol thus has truth to it.
Testosterone is a cortisol antagonist. Testosterone and cortisol are in a constant battle for the activity of LPL, the enzyme that prepares fat cells for storage. Cortisol stimulates LPL and testosterone inhibits it. This way, testosterone has a permissive effect on cortisol’s action. Cortisol will pile fat on your belly only if there is not enough testosterone to stop it.
Estrogen
In men, the effect of estrogen is naturally incorporated into the effect of testosterone. Estrogen production (estradiol + estrone + estriol) is always proportional to androgen production in healthy men. The main producer of estrogen is the enzyme aromatase. Aromatase converts a portion of androgens to estrogens. Most estrogen comes from the aromatase of testosterone to estradiol. The full biosynthesis pathway of estrogen by aromatase from adrogens is shown in the image below.
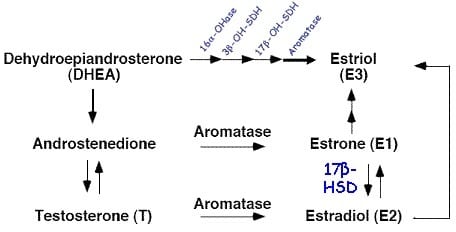
Either way, estrogen is always a proportion of testosterone. They rise together, they fall together. As such, it is impossible to distinguish between the fat storing effects of testosterone and estrogen in healthy men. This again conflicts with BioSignature Modulation.
The female perspective
What testosterone is to men, estrogen is to women. In women, estrogen is the primary cortisol antagonist that reduces LPL activity. This is a dose-response effect, not just a permissive effect like that of testosterone in men. A woman with high estrogen levels will store fat in the pear shape pattern. A woman with low estrogen levels will have a more evenly spread fat distribution pattern with more fat on the abdomen and torso and less on the lower body. So, the BioSignature claim that high estrogen is associated with fat storage on the lower body is true in women (only). However, you could just as well say estrogen is associated with a lack of fat storage on the abdomen [2,3].
Testosterone in women has the opposite effect of estrogen and therefore centralizes fat storage just like cortisol. The exact mechanisms are not yet known, but we can readily observe the effect of increasing testosterone and decreasing estrogen during menopause. Fat shifts from the lower to the upper body. The same occurs in woman-to-man transsexuals undergoing testosterone therapy: the fat distribution pattern becomes more masculine (pear to apple transformation?). Estrogen prevents this effect of testosterone by decreasing androgen receptor density. Estrogen also stimulates growth hormone production [2, 3].
Shrinking fat with growth hormone
Contrary to what its name suggests, growth hormone has a shrinking effect on fat cells (lipolysis). Growth hormone is also a cortisol antagonist and it increases the anti-cortisol activity of testosterone in men and estrogen in women. High growth hormone levels will therefore preferentially burn abdominal fat, followed by fat on the torso and lastly fat on the lower body.
In Poliquin’s BioSignature Modulation, the calves and knee are the sites that determine a person’s growth hormone level. This practice was developed in 1981 by a group of doctors from Emory University. They developed a Z-formula based on skinfold thickness measurements that could diagnose growth hormone deficiency with over 90% certainty. But there’s a catch. Two catches actually.
First, the fat distribution associated with growth hormone deficiency was exactly as you’d expect if you understood all of the above: the centralized fat storage pattern also associated with high cortisol. The skinfold thickness sites that were higher in individuals with low growth hormone production than in healthy individuals were the abdomen (suprailiac, abdominal low and high), chest (pectoral) and back (subscapular). The knees and calves were only the 4th and 5th best predictors. So, this fat storage pattern could be associated with not just growth hormone deficiency but also high cortisol or, in women, low estrogen or high testosterone. The researchers only made the connection to growth hormone deficiency because this was a study of children with short stature. Height, i.e. shortness, was the number one predictor of low growth hormone production and only the combination of short stature and a centralized fat storage pattern could identify growth hormone deficient children.
You know these disclaimers that make you question why the hell they even need to put them up there, like “No surfing on the train” or “Unsuitable to dry animals” on a microwave? The researchers of the above study also put one in there: “the particular formula developed here is not recommended for general use”.
And it gets worse. As you’d expect in a centralized fat storage pattern, children with growth hormone deficiency had lower knee and calf skinfold thickness measurements than healthy controls. This is the complete opposite of Poliquin’s BioSignature prediction.
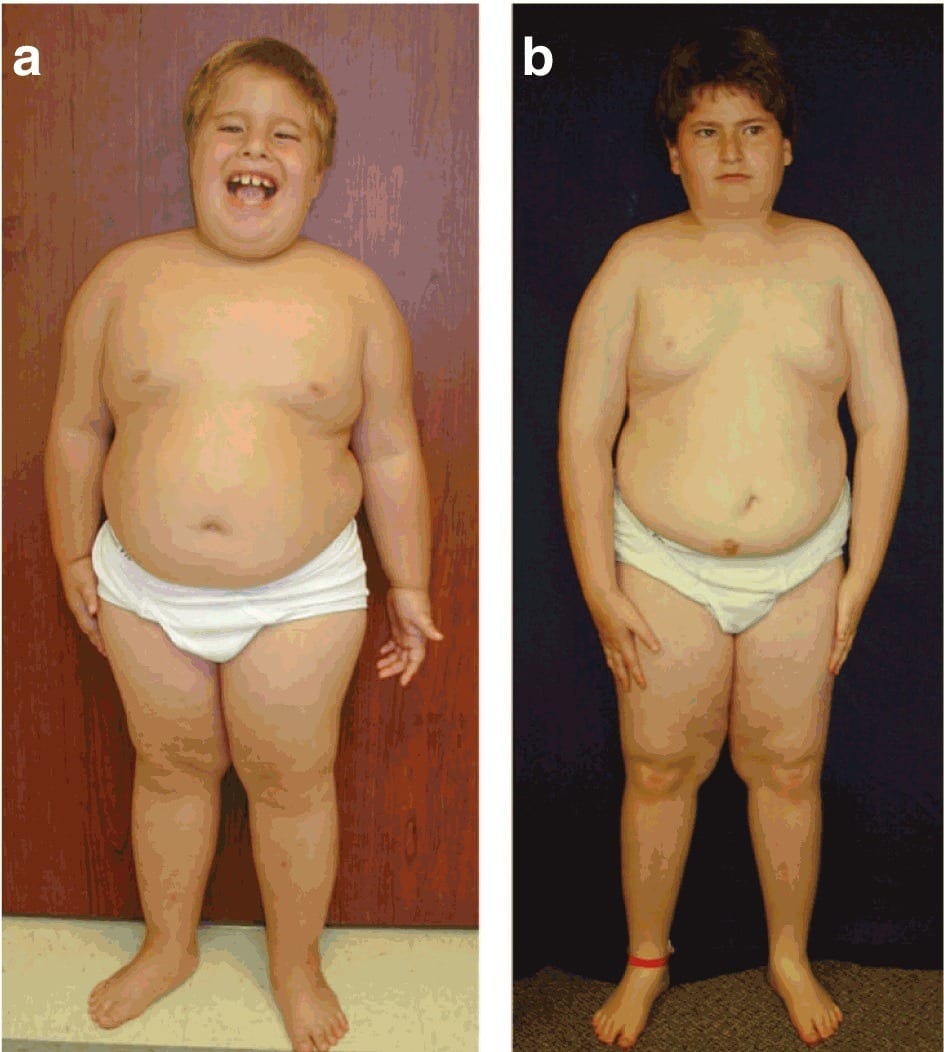 Growth hormone deficient children are not a representative sample population for fitness advice.
Growth hormone deficient children are not a representative sample population for fitness advice.
*Pictures come from the scientific journal Nature: permission was obtained to publish them.
Progesterone: the mother of complexity
Progesterone is a hormone that embodies the complexity of women. Its effects are completely dependent on the physiological state they occur in. Progesterone stimulates LPL and can thus increase fat storage, but it also blocks the glucocorticoid receptor and thus decreases the fat centralizing effects of cortisol [2].
To make matters more complex, progesterone reduces the aromatization of testosterone to estrogen. More progesterone thus generally means less estrogen, especially in men. Yet less estrogen can also reduce the effects of progesterone itself, because estrogen and progesterone strongly interact via cross-talk at both the estrogen and the progesterone receptor sites [2, 3].
This complexity is probably the reason why progesterone is not featured in BioSignature Modulation, even though it can have strong effects on regional fat storage.
Thyroid hormone: pumping up the pseudoscience
Poliquin’s BioSignature Modulation states that excessive fat storage on the ribcage (midaxillary skinfold) indicates poor thyroid function. Research has found the opposite: central obesity is positively related to thyroid activity.
Specifically, people with a lot of fat on their midsection have significantly higher concentrations of the most active thyroid hormone: free T3 (triiodothyronine). There are several mechanisms by which thyroid activity increases in response to obesity. For example, leptin, a hormone produced by fat cells, stimulates the conversion of T4 (thyroxine) to the more active T3 by altering the activity of deiodinases. Additionally, leptin sets the entire production chain of thyroid hormone in motion by increasing the transcription of pro thyrotropin-releasing hormone (TRH) [2].
More generally, thyroid hormone production is linearly related to total body weight. The fatter you are, the more active your thyroid is. If you think about it, this makes perfect sense. The bigger you are, the higher your metabolism has to be and your thyroid is the key regulator of your metabolism [2].
The change in thyroid function and consequently metabolism is also a thermo-adaptive response with an evolutionary purpose. If you lose weight, thyroid activity goes down to decrease your energy expenditure and prevent you from starving. If you gain weight, thyroid activity goes up to prevent you from becoming obese. If the evolutionary benefit of staying lean is not immediately obvious to you, consider that obesity is linked to virtually every pathology and disease state known to man, including infertility [2, 3].
Insulin: the truth under a layer of fat
Insulin is often called the storage hormone. And with good reason. Insulin interacts strongly with LPL to store fatty acids from the blood in fat tissue. Since cortisol stimulates the production of LPL, insulin reinforces the effect of cortisol. High insulin levels can therefore lead to preferential fat storage around the midsection [2].
So BioSignature’s claim that insulin levels correspond with fat storage on the love handles and back is not too far off from the truth. Both sites are part of your midsection after all. The choice for these specific sites was probably based on old research. Back in the good old days, before today’s youth depraved our society, the triceps skinfold was used to estimate a person’s fat percentage. This is what doctors call a non-invasive site and the back of the arm generally stores enough fat to pinch. The alternative? Christian chick visits the doctor’s office. Creepy doctor tells her to take off her dress to measure her ‘umbilical skinfold’. #awkward. When research showed the subscapular skinfold was a better predictor of insulin problems, the doctors were obviously overcome with joy. “Now we can play the diabetes card. Perhaps we can even take the bra off!” Subsequent research showed that the relation between insulin sensitivity and back fat disappears virtually completely when abdominal fat is also taken into account. High insulin levels are associated with centralized fat storage after all and the subscapular skinfold on the back is more centrally located than the triceps but less centrally located than the abdomen.
Summary: hormonal regulation of regional fat storage
How hormones affect where you store fat is relatively easy to summarize. Men genetically store fat pretty evenly spread over the body, giving them a ‘banana shape’. Women naturally have a pear shape where fat is preferentially stored on the hips and legs. The natural figure of both genders is only altered in one way by hormones: cortisol centralizes fat storage and changes the fat storage pattern to an apple shape. Other hormones merely change the strength of cortisol’s effect. Hormones do not affect fat storage only on specific sites. They centralize or decentralize the entire body’s fat storage pattern.
The hormonal regulation of regional fat storage in men is summarized in the following picture. The straight arrows indicate stimulatory effects. The broken arrows indicate inhibitory effects. Visceral adipose tissue represents a central fat storage pattern with lots of fat on the midsection, less on the rest of the torso and least on the lower body. In women, estrogen, progesterone and testosterone have different effects as discussed.
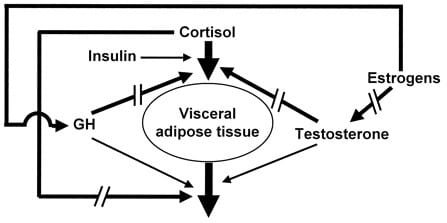
BioSignature vs. science
The scientific research differs greatly from Poliquin’s BioSignature Modulation, where every hormone has a different square inch of body space where it supposedly directs all fat storage. BioSignature doesn’t take into account the large gender differences in the hormonal effects of testosterone, estrogen and progesterone – the latter isn’t taken into account in the first place. Nor does BioSignature Modulation consider the strong interaction effects between all the hormones at play. The following table summarizes the discrepancies between BioSignature Modulation and scientific research.
| Hormone | BioSignature says | Science says |
| Testosterone | Affects fat storage on the chest and triceps | Decentralizes fat distribution in men, conditional on cortisol concentration; mild centralization in women |
| Estrogen | Affects fat storage on the hips and thighs | Suppresses testosterone in men; decentralizes fat distribution in women |
| Cortisol | Affects fat storage on the stomach | Centralizes fat distribution |
| Growth hormone | Affects fat storage on the knees and calves | Decentralizes fat distribution |
| Progesterone | Nothing | Decentralizes fat storage, conditional on estrogen and cortisol concentrations |
| Thyroid hormone | Affects fat storage on the ribcage | Doesn’t affect fat distribution |
| Insulin | Affects fat storage on love handles and back | Reinforces the effect of cortisol |
The other way around
BioSignature Modulation is wrong on many accounts of how hormones affect where the body stores fat. It is wrong on a much more fundamental level as well. Fat has a much stronger effect on hormones than hormones have on fat! We touched on this earlier. Fat regulates your thyroid function, but thyroid hormone doesn’t affect where you store fat. And fat does a lot more than regulating your thyroid.
Zooming in on fat
Contrary to what the average lay person thinks, fat tissue is not just a repulsive storage site. Fat tissue is actually very complex and scientists are still figuring out how different adipocytes function. For example, beige fat cells with a distinct physiology have only recently been identified though brown fat cells were already identified in the 16th century.
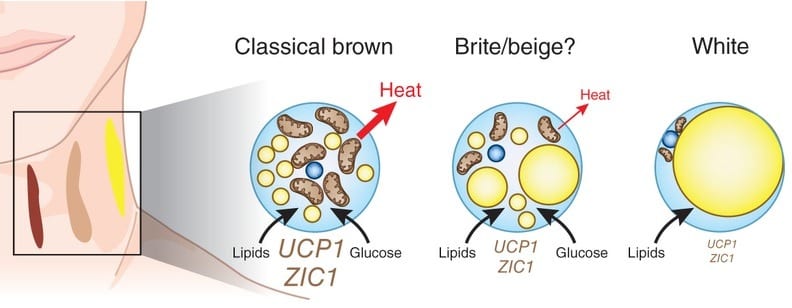
More importantly, fat tissue is far from inactive. Fat cells have their own vascular circulation, nerve cells and immune cells. Fat tissue is metabolically active and functions as an important endocrine organ. It regulates the secretion of many hormones, growth factors and enzymes, including the following [2].
- Adiponectin – regulates your blood sugar
- Resistin – regulates the build-up of cholesterol in your arteries
- Adipokines – regulate your immune system
- PAI-1 (you don’t want to know what it stands for) – regulates blood clotting
- Leptin – regulates your metabolism and appetite
- TNF and interleukins – regulate inflammation
- IGF-1 – regulates cell division and growth
- Aromatase – converts androgens like testosterone to estrogens
- Estrogen – after menopause women rely on fat tissue for up to 100% of their estrogen production
- Testosterone – about half of women’s testosterone production is derived from fat tissue
Vicious visceral fat and diabetes
As you can see, fat tissue is actively involved in the regulation of most of the important systems in your body. Of specific interest here is that fat’s function and composition varies depending on its location in the body.
Importantly, the visceral fat depot in your belly is directly connected to your liver by the hepatic portal vein. Visceral fat is highly metabolically active and has a high turnover, so if you have a large visceral fat depot AKA a gut, your liver is flooded with free fatty acids. Your liver then has to compensate by clearing less insulin from your body. As a result, insulin levels are chronically elevated. This process is further reinforced by the production of TNF and interleukins in fat tissue, especially visceral fat tissue, that cause inflammation and insulin resistance. This in turn reduces adiponectin gene expression, which messes up your blood sugar regulation even further. This spiral of obesity ends in type 2 diabetes [2].
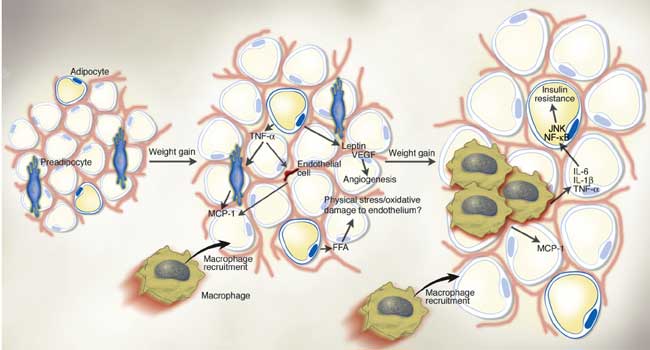
Obesity: king of disease
It gets worse still. The chronically elevated insulin levels stimulate the aromatase of testosterone to estrogen. Fat tissue also intrinsically produces a large amount of testosterone in women and estrogen in men. The result is that the hormonal profile of fat men starts resembling that of women: higher estrogen, lower testosterone. Conversely, fat women produce more testosterone [2].
These hormonal profiles hyperactivate the hypothalamic-pituitary axis. Essentially, your brain loses control over your hormonal balance, which is why obese women often menstruate irregularly. The overly sensitive hypothalamic-pituitary axis responds excessively to stress and pumps up cortisol production. Growth hormone production is suppressed accordingly [2].
If you understood how hormones regulate fat distribution, you’ll see that obesity causes a very one-sided hormonal profile for your fat distribution. All the hormones that decentralize your fat distribution are suppressed and all the hormones that centralize your fat distribution are stimulated. Obesity thus causes both genders to invariably develop an apple shaped fat storage pattern and results in the Metabolic Syndrome.
Fat is the cause, not the result
Most importantly, the causal mechanisms that link obesity to hormonal deregulation are much stronger than the effects of hormones on where you store fat. Baseline insulin action in healthy individuals only minimally affects where you store fat, but obesity inevitably causes insulin resistance. A healthy body has extremely meticulous homeostatic machinery in place that regulates its systems, like your level of blood glucose. This is why the insulin and glycemic index of your food is irrelevant if you’re healthy and a carb is a carb.
The same is true for the other hormones. Only serious clinical pathologies and states of disease where your body’s natural feedback mechanisms are disrupted, like obesity, considerably affect where you store fat. Think in terms of menopause (decrease in estrogen, increase in testosterone), Cushing’s syndrome (excessive cortisol), acromegaly (excessive growth hormone) and hormonal sex-changes. The effects of minor changes in your diet, training and supplementation have a negligible effect on where you store fat if you’re already lean and healthy [2].
BioSignature’s self-fulfilling prophecy
Then how come BioSignature ‘works’? The internet is filled with before-after photos and testimonials. Aside from before-after photos and testimonials being highly susceptible to manipulation, BioSignature’s success is very easy to explain.
Take any fat individual. Now randomly pick a skinfold and the associated hormone and tell the person that hormone is the cause of his or her fat distribution and should be corrected. You don’t even really need to do the caliper testing. You can tell an overweight male they have low testosterone, high estrogen, low growth hormone, high insulin or high cortisol based on any skinfold reading being high. You’ll always be right because all the skinfolds are thick and this hormonal profile is caused by being overweight in the first place.
Now you give them generic weight loss advice and, as a bonus for convincing them they have a problem they didn’t know they had, you get to sell them tons of supplements to fix the problem.
In the case of thyroid hormone or sex hormones in women, you may be dead wrong in your prediction of the serum hormone concentration. But that doesn’t matter either. Because the solution is always weight loss and weight loss will always decrease all the skinfold readings. Afterwards, you can always say “See: we just had to correct your hormonal imbalance”. It’s a self-fulfilling prophecy.
Guy Droog and I asked several certified BioSignature and Personal Hormonal Profiling practitioners how many people taking the caliper test get the result “Congratulations, all your serum hormone concentrations are within the physiological reference range. You could lose some fat, but this has nothing to do with your hormones.” Zip, zilch, zero, according to Mitchell from Undisputed and one trainer who wishes to remain anonymous. “No comment” the others responded.
The verdict on Poliquin’s BioSignature Modulation
Being fat is unhealthy and unaesthetic. Weight loss is the solution. It’s that simple. But that truth is not sexy, cannot be marketed. People want to pinpoint something specific as the cause of their weight, not get told they have to eat less. People want to hear there’s a new program with a great story and (pseudo) scientific techniques that will make everything better without going on a traditional diet. If this program so happens to sell a ton of supplements along with it, all the better. Success in a pill is always easier to swallow than the hard truth.
Let this be the simple message of a complex story. Be realistic about your body and don’t be deceived by marketing and sales pitches. You don’t need to buy a truckload of supplements to look great and be healthy. If you have genuine cause to believe you have a hormonal imbalance, have blood work done. If you want help in reaching your fitness goals, hire a personal trainer who knows what he’s talking about instead of a brainwashed Poliquin disciple that fills in your skinfold readings in a piece of software that spits out a generic program and a grocery list of supplements. Base your fitness program on real science instead of a fairy tale and your dream physique will become a reality.
 Want more content like this?
Want more content like this?
Then get our free mini-course on muscle building, fat loss and strength.
By filling in your details you consent with our privacy policy and the way we handle your personal data.

